

Strategic Way to Develop Your Career
Embark on a transformative learning journey with our Graduate Certificate Programmes, to gain a holistic perspective of the regulatory landscape, strengthen your expertise, and advance your career goals at your own pace.
Leading the way in healthcare education, our Graduate Certificate Programmes are designed to go beyond traditional knowledge transfer. Guided by the Duke-NUS and CoRE education philosophy, our programmes are designed to impart the latest scientific and regulatory knowledge and critical skills through practical, and application-based learning. As advocates for continuous learning, we are thrilled to support you in gaining the necessary expertise, abilities and competencies required to excel in the dynamic regulatory ecosystem.
Key features of the programme include:
- Strengthening CoRE Regulatory Skills: Building a solid foundation in regulatory principles for informed decision-making and supporting innovation.
- Gaining Interdisciplinary Competencies: Exploring the technical, scientific, and ethical aspects essential for advancing safe and effective healthcare products.
- Engaging in Real-World Application: Participating in hands-on learning to tackle complex challenges in regulatory science.
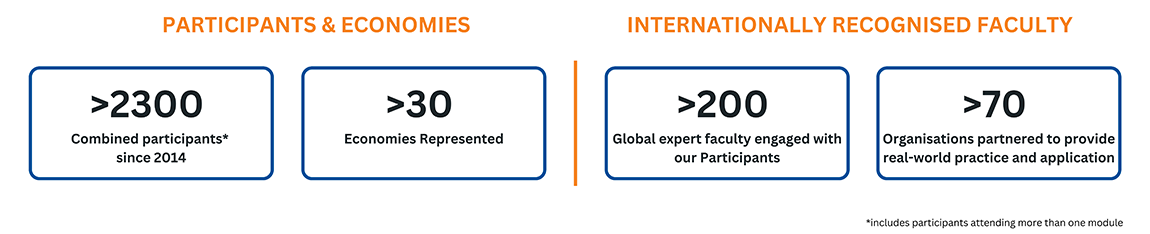
Our Impact
Crafted for early and mid-career professionals in regulatory roles within both the public and private sectors, our programmes are highly beneficial for healthcare professionals—including biomedical researchers, product developers, and policymakers—who are eager to understand the critical role of regulatory science in shaping the broader healthcare landscape.

Asia's First Academic Regulatory Training Programmes
Designed with your professional commitments in mind, the flexible curriculum allows you to tailor your learning journey to fit your career goals, while accommodating your busy work schedule.
Delivered through a blended learning approach, the programme structure combines an online learning component prior to the 5-day in-person workshop. This structure ensures both; independent learning as well as a rich interactive experience tailored to accommodate the demands of your full-time role.
To earn a Graduate Certificate, you are required to successfully complete the four below courses within your preferred Graduate Certificate programme. For added flexibility, you may choose to enroll in an individual standalone course, allowing you to focus on your specific areas of interest. This adaptable framework enables you to chart your unique path in career development.

For more information or registration, please click HERE or contact us at: lifelonglearning@duke-nus.edu.sg
